-
Chemical bonding and Molecular structure
Ionic Bond or Electrovalent Bond
An ion is an atom or group of atoms which has acquired charge due to the loss or gain of one or more electrons. When an atom gains an electron to form a negative ion (anion), it will increase in size. On the other hand, when an atom loses an electron to give positive ion (cation), it will contract. The electron lost or gained is always from the outermost shell.
When two atoms, one of which can lose one or more electrons to attain a noble gas configuration and the other can receive these electrons and thereby acquire a noble gas configuration, they are said to be bonded by an ionic bond. Since the loss and gain of electrons by atoms results in the formation of ions, ionic bond is formed when two ions of opposite charge interact with each other and are thus held together by electrostatic or coloumbic attraction. The formation of potassium chloride (KCl), is illustrated below.
\(\dot{K}(1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{1}})\underset{1\,\,electron}{\mathop{\xrightarrow{loses}}}\,\overset{+}{\mathop{K}}\,\underset{(Ar\,\,configuration)}{\mathop{(1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}})}}\,\)
\((1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}4{{s}^{1}})\underset{1\,\,electron}{\mathop{\xrightarrow{gains}}}\,{\mathop{Cl^-}}\,\underset{(Ar\,\,configuration)}{\mathop{(1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}})}}\,\)
From the above illustrations, it is clear that the formation of an ionic compound is obviously related to the ease of formation of the cations and anions from the neutral atom, which depends on two main factors:
i) Ionization energy: Lower the value of ionization energy of an atom, greater will be the ease of formation of the cation from it.
ii) Electron affinity: Higher the electron affinity of an atom, greater the ease of formation of the anion from it.
Lattice Energy
When one mole of an ionic solid is formed from its constituent gaseous ions, the energy released is called the lattice energy.
Energetics of Formation of Ionic Substances: The energy included in the formation of an ionic compound from its constituent elements may be considered as shown by the Born-Haber Cycle for the formation of one mole of sodium chloride from sodium and chlorine.
\(N{{a}_{(s)}}\underset{(+S)}{\mathop{\xrightarrow{Sub\lim ation}}}\,N{{a}_{(g)}}\underset{(+1)}{\mathop{\xrightarrow{Ionisation}}}\,Na_{(g)}^{+}+{{e}^{-}}\)
\(\frac{1}{2}C{{l}_{2(g)}}\underset{(+1/2D*)}{\mathop{\xrightarrow{Dissociation}}}\,C{{l}_{(g)}}\underset{(-{{E}_{A}})}{\mathop{\xrightarrow{Addition\,\,of\,\,{{e}^{-}}}}}\,Cl_{(g)}^{-}\)
Na+(g)+Cl-(g)
NaCl(s)
Where S = heat of sublimation of sodium metal
I = ionization energy of sodium
D = heat of dissociation of molecular chlorine
EA = electron affinity of chlorine, and
U = lattice energy of sodium chloride
The amount of heat liberated in the overall reaction is the heat of formation of sodium chloride. From the above
\(\Delta H=S+I+\frac{1}{2}D-{{E}_{A}}-U\)
The most important of these energy terms are I, EA and U, since these are considerably greater than the remaining terms S and D.
More the negative value of the heat of formation, greater would be the stability of the ionic compound produced. Thus on the basis of the above equation, formation of an ionic compound is favored by
a) low ionization energy (I) of the metal.
b) high electron affinity (EA) of the other element.
c) higher lattice energy (U) of the resulting compound.
Formation of Ions with Higher Charges: Formation of a cation with unit positive charge is easy if the first ionization energy is low as in the case of alkali metals. Alkaline earth metals ionizes in two successive steps.
Eg : Mg \(\xrightarrow{{}}\) Mg+ + e–
Mg+ \(\xrightarrow{{}}\) Mg2+ + e–
But energy needed to ionize alkaline earth metals is higher than alkali metals.
However, dipositive ions like Mg2+, Ca2+, Sr2+ and Ba2+ are quite common. Formation of a tripositive ion like Al3+ requires much more energy (5138 kJ) which is not available ordinarily. Successive ionization energies of aluminum are:
\(Al\xrightarrow{{{E}_{1}}}A{{l}^{+}}+{{e}^{-}}\) E1 = 577kJ
\(A{{l}^{+}}\xrightarrow{{{E}_{2}}}A{{l}^{2+}}+{{e}^{-}}\) E2 = 1816kJ
\(A{{l}^{2+}}\xrightarrow{{{E}_{3}}}A{{l}^{3+}}+{{e}^{-}}\)E3 = 2745kJ
It is on this account that most of aluminum compounds are covalent. In solution, however, aluminum is known to give hydrated ions \({{[Al{{({{H}_{2}}O)}_{6}}]}^{3+}}\). This is possible because of the high heat of hydration of \(A{{l}^{3+}}\) The energy liberated during hydration of ions is sufficient for ionization.
Similarly, anions with unit negative charge \(O-O\) are very common. This is because the electron affinity of these atoms is negative and quite high. Formation of anions carrying two units of negative charge \((e.g.\,\,{{S}^{2-}},\,\,{{O}^{2-}})\) is not so easy as their electron affinities are negative i.e., energy is needed to add second electron. Formation of anions carrying three units of negative charge \((e.g.\,\,{{N}^{3-}},\,\,{{P}^{3-}})\) is almost rare.
Characteristics of Electrovalent Compounds
Melting and Boiling Point: Due to the strong electrostatic force between the ions in a crystal of an electrovalent compound. The energy needed to overcome these forces and break down the crystal lattice. Hence such compounds possess high melting and boiling points.
Electrical Conductivity: When an electrovalent compound is molten or dissolved in a solvent of high dielectric constant e.g., water, the binding forces in the crystal lattice disappear and the component ions become mobile. Under the influence of applied electrical field, the ions get charged and thus act as charge carrier of the current. Hence their molten forms or solutions conduct electricity.
Solubility: Ionic compounds are soluble in polar solvents like water, because of molecules of the polar solvent interact strongly with the ions of the crystal and the solvation energy is sufficient to overcome the attraction between the ions in the crystal lattice. Dissolution is also favoured by the high dielectric constant of the solvents such as water, since this weakens the interionic attractions in the resulting solutions.
Non-polar solvents like benzene and carbon tetrachloride do not solvate the ions as their dielectric constants are low. Ionic compounds are, therefore insoluble in non-polar solvents.
Ionic compounds like sulphates and phosphates of barium and strontium are insoluble in water (because lattice energy is greater than hydration energy). This can be attributed to the high lattice energies of these compounds due to polyvalent nature of both the cation and the anion. In these cases, hydration of ions fails to liberate sufficient energy to offset the lattice energy.
Illustration :
Which one is correct for ionic bond
(a) it is nondirectional
(b) it is formed by the elements with same electronegativity
(c) it is formed by overlapping of orbitals
(d) Both (a) and (c) are correct
Sol.(a) Ionic bond is non directional and covalent bond is directional . It is formed by the element with different electronegativities.Ionic bond is formed due to attraction between positive and negative ions and not due to over lapping.
Illustration :
Which one having highest hydration energy
(a) Na+ (b) Li+ (c) Cs + (d) K+
Sol.(b) Li+ ion has highest hydration energy dute to small size
-
Explanation of Ionic or Electrovalent Bond
Explanation of Ionic or Electrovalent Bond
From the Kössel and Lewis treatment of the formation of an ionic bond, it follows that the formation of ionic compounds would primarily depend upon:
1. The ease of formation of the positive and negative ions from the respective neutral atoms;
2. The arrangement of the positive and negative ions in the solid, that is, the lattice of the crystalline compound.
The formation of a positive ion involves ionization, i.e., removal of electron(s) from the neutral atom and that of the negative ion involves the addition of electron(s) to the neutral atom.
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaamytamaabm % aabaGaam4zaaGaayjkaiaawMcaaiabgkziUkaad2eadaahaaWcbeqa % aiabgUcaRaaakmaabmaabaGaam4zaaGaayjkaiaawMcaaiabgUcaRi % aadwgadaahaaWcbeqaaiabgkHiTaaakiaacUdaaaa!4339! M\left( g \right) \to {M^ + }\left( g \right) + {e^ - };\)
Ionization enthalpy
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaamiwamaabm % aabaGaam4zaaGaayjkaiaawMcaaiabgUcaRiaadwgadaahaaWcbeqa % aiabgkHiTaaakiabgkziUkaadIfacqGHsisldaqadaqaaiaadEgaai % aawIcacaGLPaaacaGG7aaaaa!4323! X\left( g \right) + {e^ - } \to X - \left( g \right);\)
Electron gain enthalpy
\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaamytamaaCa % aaleqabaGaey4kaScaaOWaaeWaaeaacaWGNbaacaGLOaGaayzkaaGa % ey4kaSIaamiwaiabgkHiTiabgkziUkaad2eacaWGybWaaeWaaeaaca % WGZbaacaGLOaGaayzkaaaaaa!431F! {M^ + }\left( g \right) + X - \to MX\left( s \right)\)
The electron gain enthalpy,\(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaeyiLdq0aaS % baaSqaaiaadwgacaWGNbaabeaakiaadIeaaaa!3A36! {\Delta _{eg}}H\),is the enthalpy change (Unit 3),when a gas phase atom in its ground state gains an electron. The electron gain process may be exothermic or endothermic. The ionization, on the other hand, is always endothermic. Electron affinity, is the negative of the energy change accompanying electron gain.
Obviously ionic bonds will be formed more easily between elements with comparatively low ionization enthalpies and elements with comparatively high negative value of electron gain enthalpy.
Most ionic compounds have cations derived from metallic elements and anions from non-metallic elements.The ammonium \(% MathType!MTEF!2!1!+- % feaagKart1ev2aaatCvAUfeBSjuyZL2yd9gzLbvyNv2CaerbuLwBLn % hiov2DGi1BTfMBaeXatLxBI9gBaerbd9wDYLwzYbItLDharqqtubsr % 4rNCHbGeaGqiVu0Je9sqqrpepC0xbbL8F4rqqrFfpeea0xe9Lq-Jc9 % vqaqpepm0xbba9pwe9Q8fs0-yqaqpepae9pg0FirpepeKkFr0xfr-x % fr-xb9adbaqaaeGaciGaaiaabeqaamaabaabaaGcbaGaamOtaiaadI % eadaqhaaWcbaGaaGinaaqaaiabgUcaRaaaaaa!3963! NH_4^ + \) ion (made up of two non- metallic elements) is an exception.It forms the cation of a number of ionic compounds.
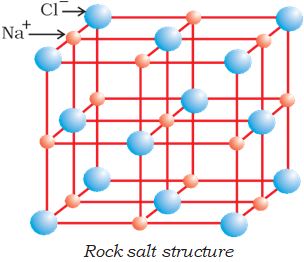
Ionic compounds in the crystalline state consist of orderly three-dimensional arrangements of cations and anions held together by coulombic interaction energies. These compounds crystallise in different crystal structures determined by the size of the ions, their packing arrangements and other factors. The crystal structure of sodium chloride, NaCl (rock salt), for example is shown below.
In ionic solids,the sum of the electron gain enthalpy and the ionization enthalpy may be positive but still the crystal structure gets stabilized due to the energy released in the formation of the crystal lattice.For example:the ionization enthalpy for Na+(g) formation from Na(g) is 495.8 kJ mol–1 ; while the electron gain enthalpy for the change Cl(g)+e– \(\to\)Cl– (g) is, – 348.7 kJ mol–1 only. The sum of the two, 147.1 kJ mol-1 is more than compensated for by the enthalpy of lattice formation of NaCl(s) (–788 kJ mol–1). Therefore, the energy released in the processes is more than the energy absorbed.Thus a qualitative measure of the stability of an ionic compound is provided by its enthalpy of lattice formation and not simply by achieving octet of electrons around the ionic species in gaseous state.
Since lattice enthalpy plays a key role in the formation of ionic compounds, it is important that we learn more about it.
-
Lattice Enthalpy
Lattice Enthalpy
The Lattice Enthalpy of an ionic solid is defined as the energy required to completely separate one mole of a solid ionic compound into gaseous constituent ions. For example, the lattice enthalpy of NaCl is 788 kJ mol–1. This means that 788 kJ of energy is required to separate one mole of solid NaCl into one mole of Na+ (g) and one mole of Cl– (g) to an infinite distance.
This process involves both the attractive forces between ions of opposite charges and the repulsive forces between ions of like charge. The solid crystal being three- dimensional; it is not possible to calculate lattice enthalpy directly from the interaction of forces of attraction and repulsion only. Factors associated with the crystal geometry have to be included.